한국응용과학기술학회 논문 검색

구리 함량 변화에 따른 Metal Corrugated Cu-Chabazite 촉매의 HC-SCR에 관한 연구
간행물 정보 : Vol. 42 No. 3, 2025년,
파일형식 : pdf





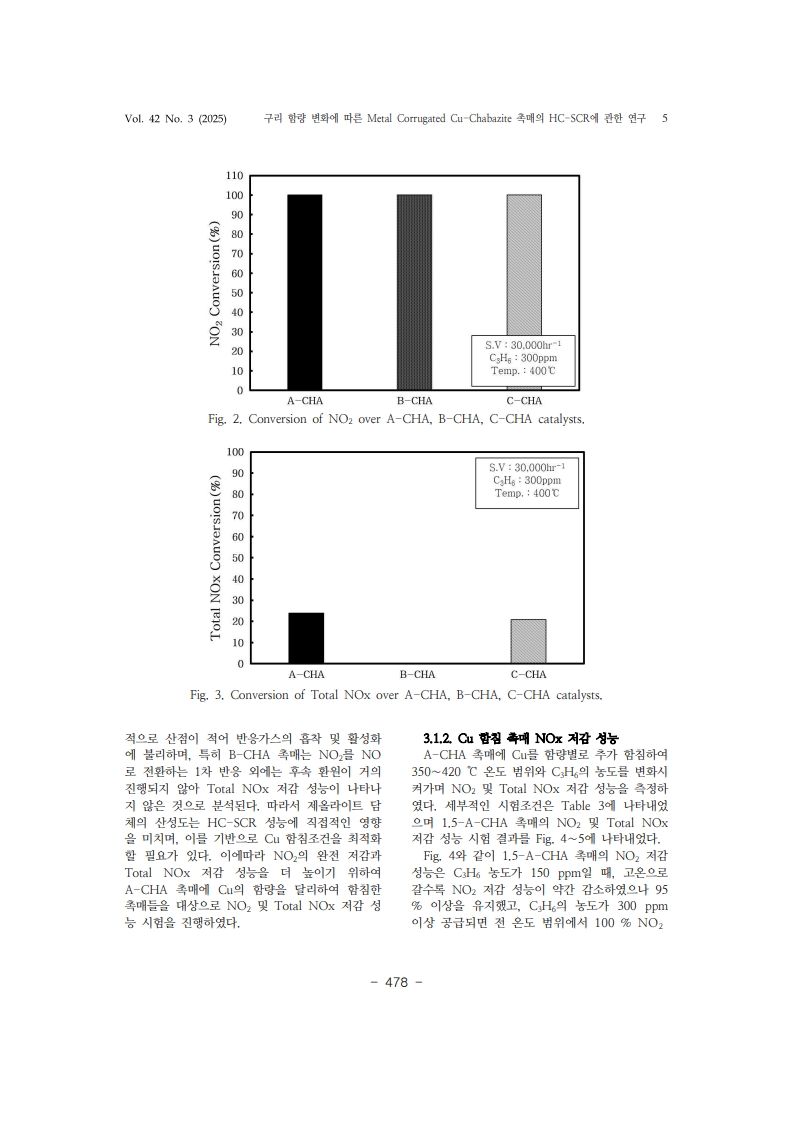
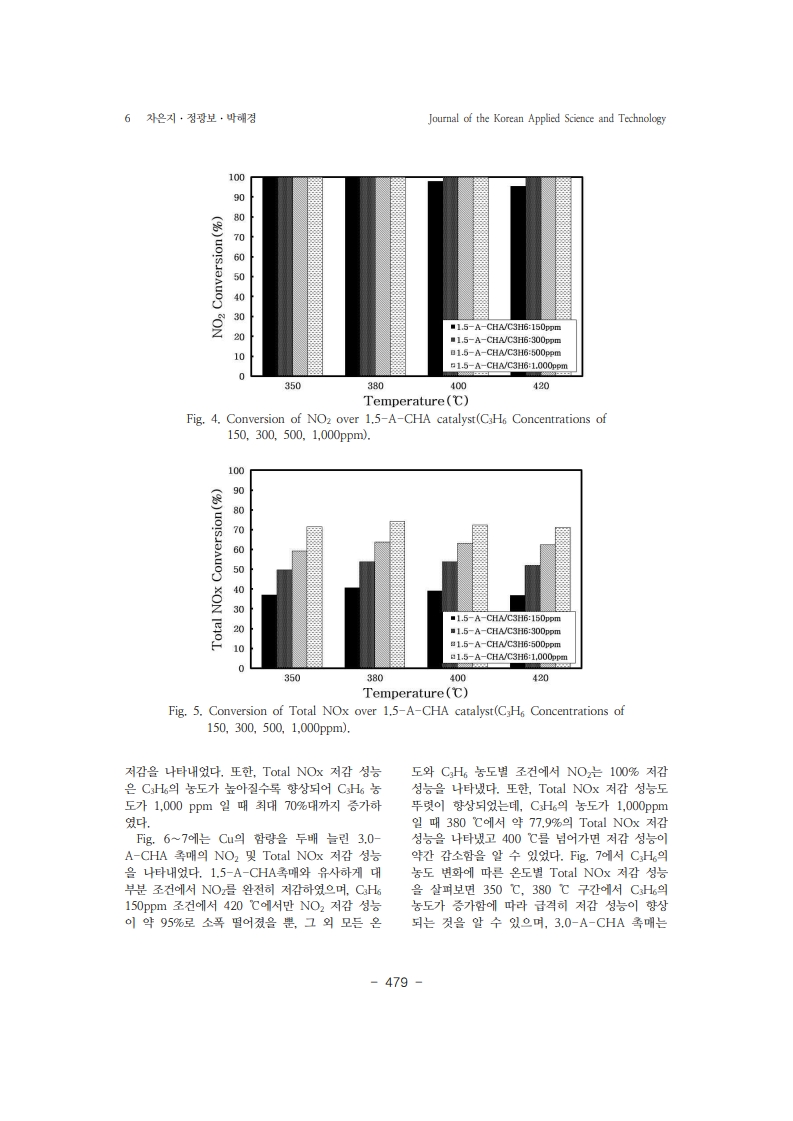
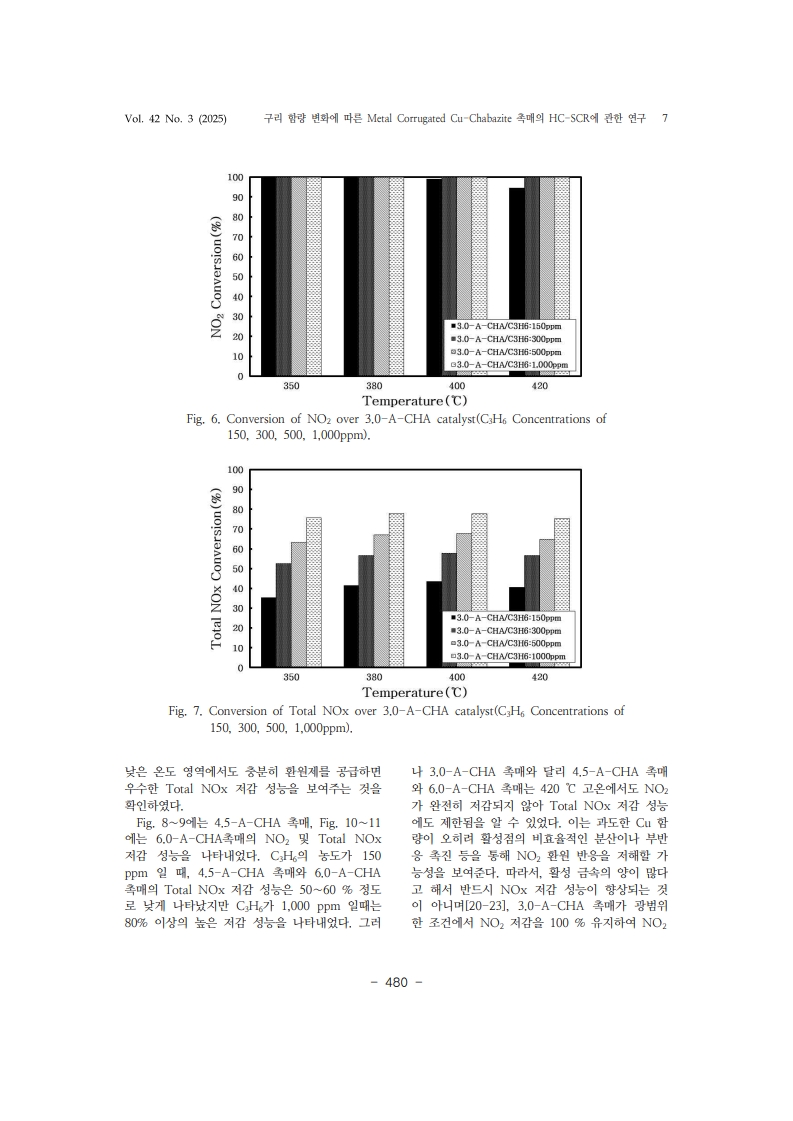
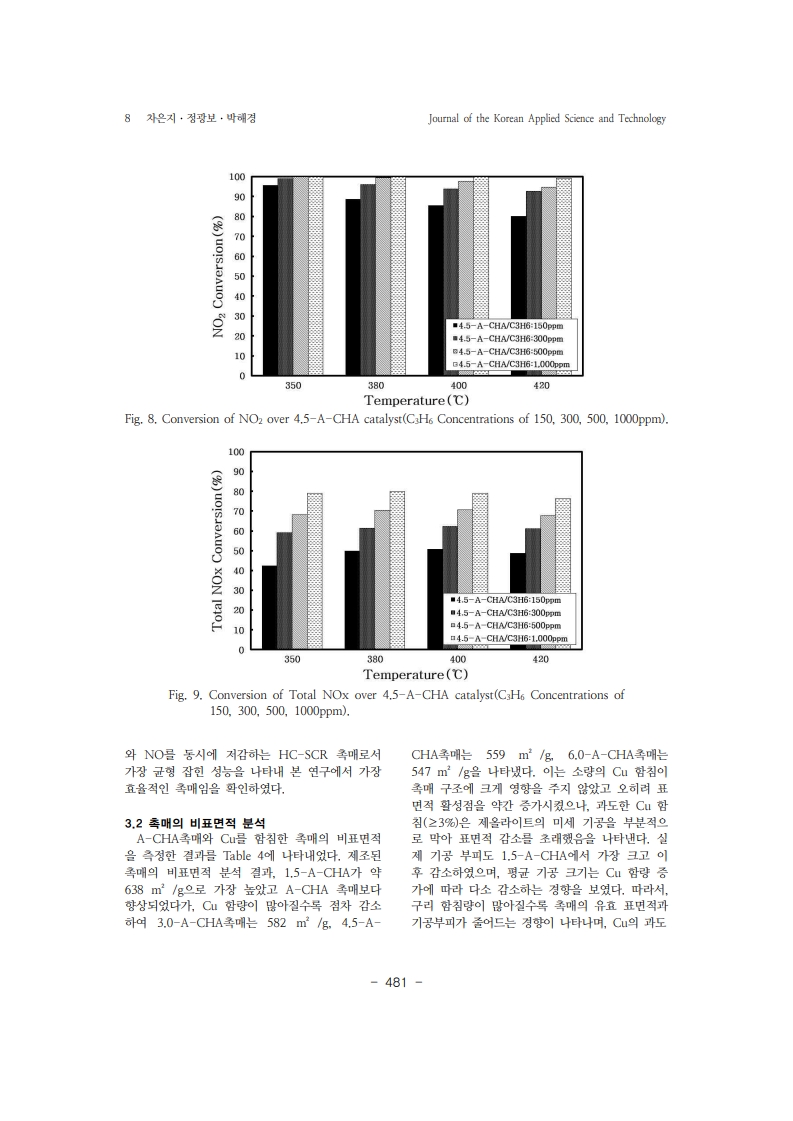



1. Kim, H. S., Kasipandi, S., Kim, J., Kang, S. H., Kim, J. H., Ryu, J. H., & Bae, J.W. “Current catalyst technology of selective catalytic reduction (SCR) for
NOx removal in South Korea.” CatalystsVol.10, No.1, pp. 52 (2020).https://doi.org/10.3390/catal10010052
2. Yang, G., Zhao, H., Luo, X., Shi, K., Zhao, H., Wang, W., & Wu, T.“Promotion effect and mechanism of the addition of Mo on the enhanced low
temperature SCR of NOx by NH3 over MnOx/γ-Al2O3 catalysts”. AppliedCatalysis B: Environmental Vol.245, pp.
743-752. (2019). https://doi.org/10.1016/j.apcatb.2018.12.080
3. Wei, L. G., Guo, R. T., Zhou, J., Qin, B., Chen, X., Bi, Z. X., & Pan, W. G.“Chemical deactivation and resistance of Mn-based SCR catalysts for NOx removal
from stationary sources”. Fuel Vol.316, pp. 123438 (2022).https://doi.org/10.1016/j.fuel.2022.123438
4. Lee, K. S., “Biological Removal of Nitrogen Oxides from Combustion FlueGases”. Appl. Chem. Eng Vol.21, NO.3, pp. 243-251 (2010).
5. Li, C., Tang, X., Yi, H., Wang, L., Cui, X., Chu, C., & Yu, Q. “Rational designof template-free MnOx-CeO2 hollow nanotube as de-NOx catalyst at lowtemperature“. Applied Surface ScienceVol.428, pp. 924-932 (2018).https://doi.org/10.1016/j.apsusc.2017.09.131
6. Lee, G. W., Shon, B. H., Yoo, J. G., Jung, J. H., & Oh, K. J. “The influence ofmixing between NH3 and NO for a De-NOx reaction in the SNCR process“.
Journal of Industrial and Engineering Chemistry Vol.14, No.4, pp. 457-467(2008). https://doi.org/10.1016/j.jiec.2008.02.013
7. Nova, I., Ciardelli, C., Tronconi, E., Chatterjee, D., & Bandl-Konrad, B. “NH3– NO/NO2 chemistry over V-based catalysts and its role in the mechanism of the Fast SCR reaction”. Catalysis Today Vol.114, No.1, pp. 3-12 (2006). https://doi.org/10.1016/j.cattod.2006.02.012
8. Chen, L., Ren, S., Liu, L., Su, B., Yang, J., Chen, Z., & Liu, Q. “Catalyticperformance over Mn-Ce catalysts for NH3-SCR of NO at low temperature:
Different zeolite supports”. Journal of Environmental Chemical Engineering Vol.10,No.2, pp. 107167 (2022). https://doi.org/10.1016/j.jece.2022.107167
9. Mrad, R., Aissat, A., Cousin, R., Courcot, D., & Siffert, S. “Catalysts for NOxselective catalytic reduction by hydrocarbons (HC-SCR)”. Applied
Catalysis A: General Vol.504, pp. 542-548 (2015).https://doi.org/10.1016/j.apcata.2014.10.021
10. Li, L., & Guan, N. “HC-SCR reaction pathways on ion exchanged ZSM-5catalysts”. Microporous and Mesoporous Materials Vol.117, No.1-2, pp. 450-457
(2009). https://doi.org/10.1016/j.micromeso.2008.07.021
11. Guo, K., Ji, J., Song, W., Sun, J., Tang, C., & Dong, L. “Conquering ammoniumbisulfate poison over low-temperature NH3-SCR catalysts: A critical review”.
Applied Catalysis B: Environmental Vol.297,pp. 120388 (2021).https://doi.org/10.1016/j.apcatb.2021.120388
12. Tronconi, E., Nova, I., Ciardelli, C., Chatterjee, D., & Weibel, M. “Redoxfeatures in the catalytic mechanism of the “standard” and “fast” NH3-SCR of NOx over a V-based catalyst investigated by dynamic methods“. Journal of Catalysis, Vol.245, No.1, pp. 1-10 (2007). https://doi.org/10.1016/j.jcat.2006.09.012
13. Sun, X., Guo, R. T., Liu, S. W., Liu, J., Pan, W. G., Shi, X., & Liu, X. Y. “Thepromoted performance of CeO2 catalyst for NH3-SCR reaction by NH3 treatment”.
Applied Surface Science Vol.462, pp. 187-193 (2018).https://doi.org/10.1016/j.apsusc.2018.08.114
14. Ihm, T. H., Jo, J. O., Hyun, Y. J., & Mok, Y. S. “Removal of Nitrogen OxidesUsing Hydrocarbon Selective Catalytic Reduction Coupled with Plasma”. Appl.
Chem. Eng Vol.27, No.1, pp. 92-100 (2016). https://doi.org/10.14478/ace.2015.1130
15. Chen, J. P., Hausladen, M. C., & Yang, R. T. “Superior catalysts for selectivecatalytic reduction of nitric oxide” (1995).
16. Traa, Y., Burger, B., & Weitkamp, J. “Zeolite-based materials for the selectivecatalytic reduction of NOx with hydrocarbons”.Microporous and Mesoporous
materials Vol.30, No.1, pp. 3-41 (1999).https://doi.org/10.1016/s1387-1811(99)00030-x
17. Sazama, P., Mokrzycki, L., Wichterlova, B., Vondrova, A., Pilar, R., Dedecek, J., & Tabor, E. “Unprecedented propane–SCRNOxactivity over template-free synthesizedAl-rich Co-BEA∗ zeolite”. Journal of catalysis Vol.332, pp. 201-211 (2015). https://doi.org/10.1016/j.jcat.2015.10.007
18. Guo, T., Pan, H., Gao, E., Zhang, X., & He, Y. “Mechanism of Propane Adsorption and the Following NO x Reduction over an In/BEA Catalyst: A Computational Study”. ACS omega Vol.7, No.5, pp.4501-4513 (2022). https://doi.org/10.1021/acsomega.1c06414
19. Orlik, S. N. “Combined effect of the redox and acid-base properties of catalystsin redox conversions of nitrogen oxides and methane”. Kinetics and Catalysis
Vol.49, pp. 537-544 (2008). https://doi.org/10.1134/s0023158408040137
20. Na, W. J., Park, H. K. “A Study on the NOx Reduction According to the SpaceVelocity Variation and Binder Content of Metal foam SCR Catalyst for
Cogeneration Power Plant Application”. The Korean Society of Applied Scienceand Technology Vol.36, No.1, pp. 153- 164 (2019)
21. Na, W. J., Park. H. K. “Metal type SCR catalyst with excellent de-NOx efficiencyat low temperature and sulfur resistance from coal combustion exhaust gas”.
International Journal of Engineering Research and Technology Vol.14, No.6,pp. 537-542 (2021).
22. Na, W. J., Park, H. K. “A Study on the de-NOx Activity of V2O5-Sb2O3/TiO2 SCR Catalyst at Low Temperature According to the Wash Coating Conditions”
International Journal of Applied EngineeringResearch Vol.13, No.15, pp. 11949-11959 (2018).
23. Na, W. J. Park, Y. J., Bang, H. S., Bang, J. S., & Park. H. K. “Effect of SO2 on NOx Removal Performance in Low Temperature Region over V2O5-Sb2O3/TiO2 SCR Catalyst Washcoated on the Metal Foam”. Clean Technol Vol.22, No.2, pp.132-138 (2016). https://doi.org/10.7464/ksct.2016.22.2.132
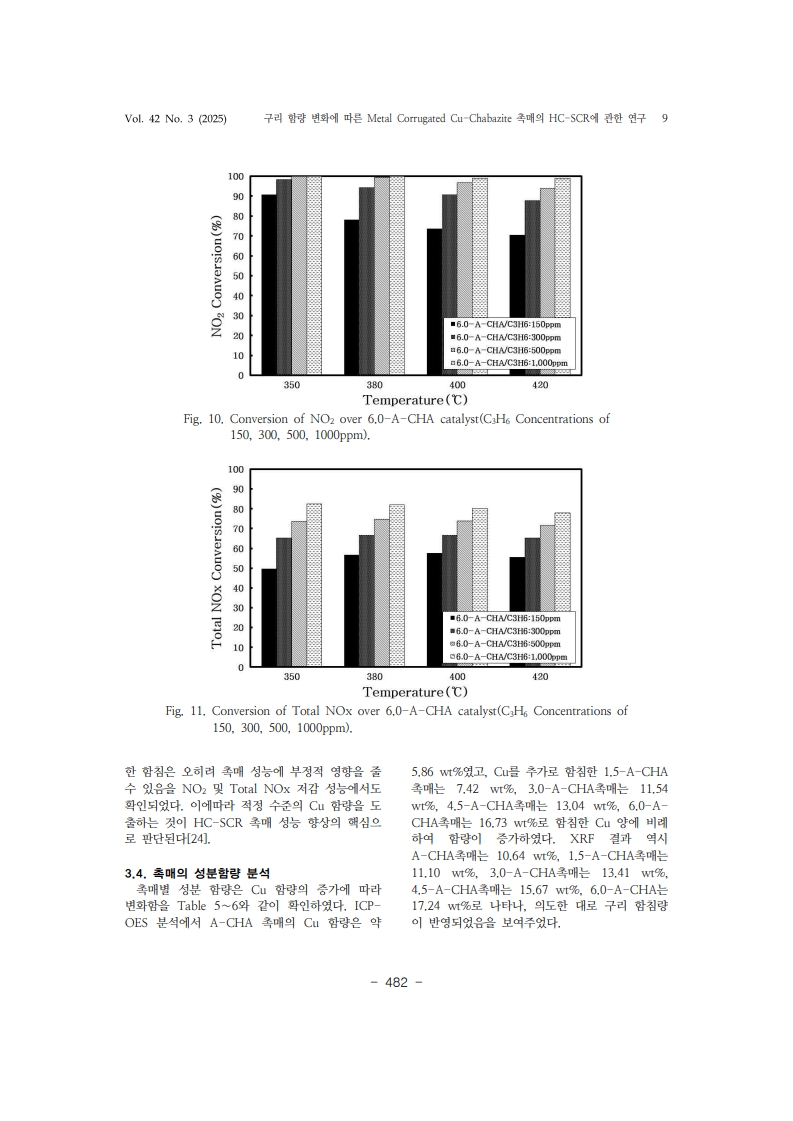
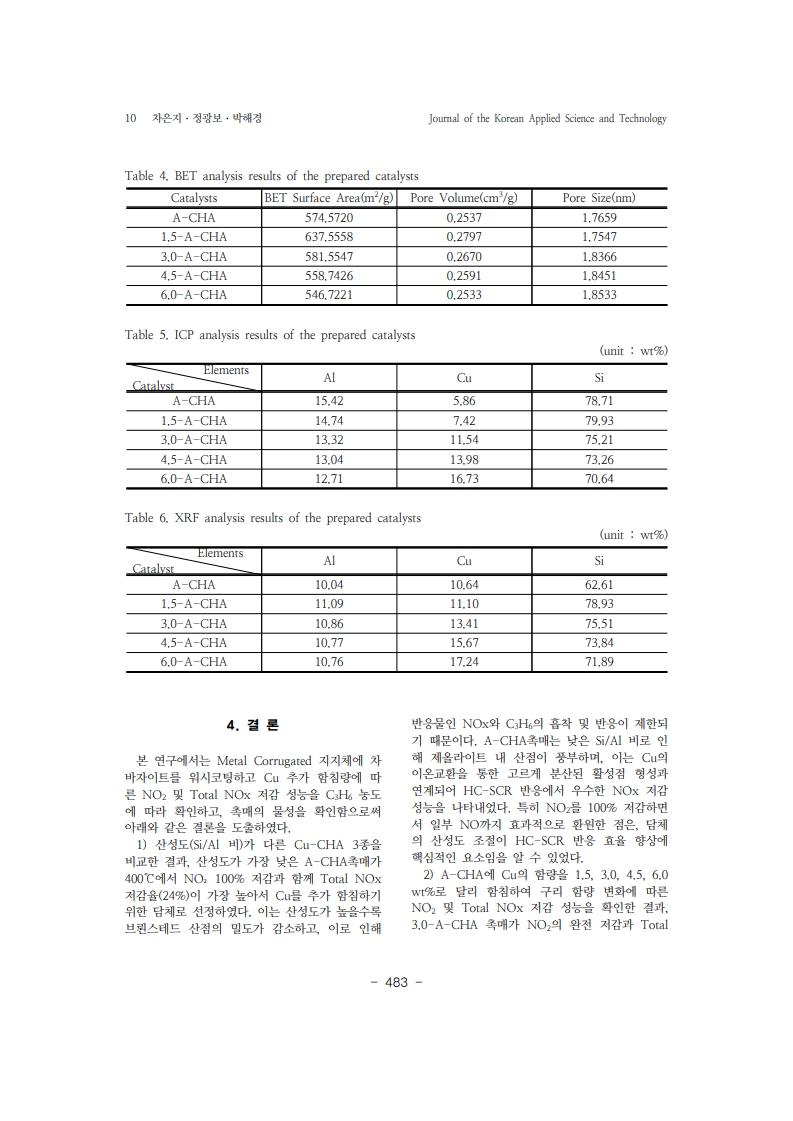
24. Lee, K., Kosaka, H., Sato, S., Yokoi, T., & Choi, B. “Effect of Cu content andzeolite framework of n-C4H10-SCR catalysts on de-NOx performances”.
Chemical Engineering Science Vol.203, pp. 28-42 (2019).https://doi.org/10.1016/j.ces.2019.03.028
